WO1993000097A1 - Pharmaceutical preparations containing torasemid - Google Patents
Pharmaceutical preparations containing torasemid Download PDFInfo
- Publication number
- WO1993000097A1 WO1993000097A1 PCT/EP1992/001437 EP9201437W WO9300097A1 WO 1993000097 A1 WO1993000097 A1 WO 1993000097A1 EP 9201437 W EP9201437 W EP 9201437W WO 9300097 A1 WO9300097 A1 WO 9300097A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- parts
- weight
- torasemide
- pharmaceutical composition
- cellulose
- Prior art date
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/20—Pills, tablets, discs, rods
- A61K9/2004—Excipients; Inactive ingredients
- A61K9/2022—Organic macromolecular compounds
- A61K9/205—Polysaccharides, e.g. alginate, gums; Cyclodextrin
- A61K9/2054—Cellulose; Cellulose derivatives, e.g. hydroxypropyl methylcellulose
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/64—Sulfonylureas, e.g. glibenclamide, tolbutamide, chlorpropamide
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/20—Pills, tablets, discs, rods
- A61K9/2004—Excipients; Inactive ingredients
- A61K9/2022—Organic macromolecular compounds
- A61K9/2027—Organic macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds, e.g. polyvinyl pyrrolidone, poly(meth)acrylates
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P7/00—Drugs for disorders of the blood or the extracellular fluid
- A61P7/10—Antioedematous agents; Diuretics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/12—Antihypertensives
Definitions
- the invention relates to torasemide-containing pharmaceutical compositions which, for. B. are effective as prophylactic or therapeutic agents against hypertension and edema, or as diuretics.
- Torasemide [chemical name: l-isopropyl-3- (4-m-toluidino-3-pyridyl) sulfonyl) urea] is e.g. B. effective as a diuretic in the context of hypertension therapy.
- Orally administered preparations containing torasemide are produced by a generally customary process by adding, for example, sugar, starch, starch derivatives, cellulose, cellulose derivatives, mold release agents, non-stick agents or other customary auxiliaries; they are applied, for example, as tablets to which lactose, corn starch, silicon oxide or magnesium stearate is added (EP-A-0,212,537).
- compositions are desired in which the storage stability of the torase itself is improved, especially when stored over a longer period of time, changes in the appearance of the preparation caused by moisture absorption are avoided, and the absorption of the torasemide is avoided is further improved by the digestive tract and which have good bioavailability. It has been found that changes in the formulations which have occurred can be caused by corn starch present in the formulation. Accordingly, this invention aims to solve such problems as are encountered in the conventional art and to make better torasemide-containing preparations available for oral administration. To solve the problems described above, numerous commonly used components, such as. B. binders and disintegrants, tests carried out.
- torasemide-containing compositions which contain hydroxypropyl cellulose or polyvinylpyrrolidone (PVP) as binders, sodium croscarmellose or crospovidone as disintegrants and customary shaping agents, such as, for. B. lactose and / or crystalline cellulose and lubricants such as. B. magnesium stearate, have excellent properties as preparations for oral administration.
- PVP polyvinylpyrrolidone
- customary shaping agents such as, for. B. lactose and / or crystalline cellulose and lubricants such as. B. magnesium stearate
- the active ingredient, the binder and the disintegrant are to be used in certain ratios to one another.
- the invention therefore relates to pharmaceutical compositions containing torasemide or a compatible salt thereof, a binder selected from the products hydroxypropyl cellulose and polyvinylpyrrolidone (PVP), a disintegrant selected from the products sodium croscarmellose, crospovidone, calcium carboxymethyl cellulose, contain low-substituted hydroxypropyl cellulose, modified starch and sodium carboxymethyl starch and conventional shaping agents and lubricants.
- PVP polyvinylpyrrolidone
- a disintegrant selected from the products sodium croscarmellose, crospovidone, calcium carboxymethyl cellulose
- torasemide preferably the active ingredient in modification 1
- the torasemide salts are not subject to any particular restriction if they are pharmacologically acceptable substances; as examples, salts with organic acids, such as. B. acetic acid, suberic acid, maleic acid, fumaric acid, malic acid, tartaric acid or methanesulfonic acid, and salts with inorganic acids, such as. As hydrochloric acid, hydrobromic acid, phosphoric acid or sulfuric acid.
- hydroxypropyl cellulose is preferred as the binder; in particular, it was found that especially those with a high degree of substitution are suitable.
- the degree of substitution of such a highly substituted hydroxypropyl cellulose is normally 50-80%, preferably 60-70%, expressed as a content (%) of hydroxypropoxyl groups.
- Commercial hydroxypropyl cellulose is used specifically.
- Polyvinylpyrrolidone is also preferred.
- Sodium croscarmellose, crospovidone, calcium carboxymethyl cellulose, low-substituted hydroxypropyl cellulose, modified starch or sodium carboxymethyl starch are preferably used as disintegrants.
- sodium croscarmellose or crospovidones are preferred.
- Sodium croscarmellose is usually understood to mean sodium carboxy ethyl cellulose, crosslinked, crosprovidone polyvinylpyrrolidone, crosslinked.
- a modified starch is preferably a starch converted to the position.
- Shaping agents or lubricants will also be added to the pharmaceutical composition according to this invention; as shaping agents are preferred z.
- the mixing ratio of the individual components in the pharmaceutical composition of this invention is normally 0.1-50 parts (here and hereinafter: parts by weight), preferably 0.5-15 parts, more preferably 1-10 parts of torasemide; 1-10 parts, preferably 2-5 parts binder; 1-20 parts, preferably 3-15 parts, more preferably 5-10 parts of disintegrant.
- the proportions of the other components that are mixed into the pharmaceutical composition of this invention are 10-150 parts, preferably 70-140 parts, more preferably 100-130 parts of shaping agents, and 0.1-5 parts, preferably 0.2-2 Parts, more preferably 0.5-1.5 parts of lubricant.
- the pharmaceutical composition of this invention is e.g. B. as a prophylactic or therapeutic against hypertension and edema, or ideally administered orally as a diuretic.
- the pharmaceutical composition of this invention is processed into a powder, granule, tablet or pellet by conventional methods.
- the application of the pharmaceutical composition according to the invention varies depending on the sex, body weight, age, disease state, etc. of the patient; Usually, a daily dose of approximately 1 to 200 mg of torasemide is administered to an adult, preferably 1 to 30 mg, divided into one to several times a day.
- 40 g torasemide were mixed with 1,064 g lactose, 32 g hydroxypropyl cellulose (content of hydroxypropoxyl groups 62%) and 24 g sodium croscarmellose, pelleted with water, dried and then sieved. 12.9 g of sodium croscarmellose and 3.2 g of magnesium stearate were mixed with 466 g of this substance and the whole was then processed into tablets by a customary process. About 2,300 tablets containing 4 mg of torasemide per tablet were produced in this way.
- the numbers indicate the weight (mg) of the ingredients in the tablet.
Abstract
Described are storage-stable pharmaceutical preparations containing torasemid as the active substance, plus a binder and a disintegrator, as well as the usual form-defining materials and lubricants.
Description
Torasemid-halticre pharmazeutische ZusammensetzungenTorasemid-halticre pharmaceutical compositions
Die Erfindung betrifft Torasemid-haltige pharmazeutische Zusammensetzungen, die z. B. als Prophylaktika bzw. Thera- peutika gegen Hypertonie und Ödeme, oder als Diuretika wirksam sind.The invention relates to torasemide-containing pharmaceutical compositions which, for. B. are effective as prophylactic or therapeutic agents against hypertension and edema, or as diuretics.
Torasemid [chemischer Name: l-Isopropyl-3-(4-m-toluidino- 3-pyridyl)sulfonyl)harnstoff] ist z. B. als Diuretikum im Rahmen einer Hypertonie-Therapie wirksam. Oral zu applizierende Präparate, die Torasemid enthalten, werden nach einem allgemein üblichen Verfahren hergestellt, indem zu dem Wirkstoff zum Beispiel Zucker, Stärke, Stärke- Derivate, Cellulose, Cellulose-Derivate, Trennmittel, Antihaftmittel oder andere übliche Hilfsstoffe zugefügt werden; sie werden beispielsweise als Tabletten, denen Lactose, Maisstärke, Siliciumoxid oder Magnesiumstearat beigemischt ist, appliziert (EP-A-0,212,537) .Torasemide [chemical name: l-isopropyl-3- (4-m-toluidino-3-pyridyl) sulfonyl) urea] is e.g. B. effective as a diuretic in the context of hypertension therapy. Orally administered preparations containing torasemide are produced by a generally customary process by adding, for example, sugar, starch, starch derivatives, cellulose, cellulose derivatives, mold release agents, non-stick agents or other customary auxiliaries; they are applied, for example, as tablets to which lactose, corn starch, silicon oxide or magnesium stearate is added (EP-A-0,212,537).
Auf der anderen Seite werden pharmazeutische Präparate gewünscht, bei denen, vor allem bei einer Lagerung über einen längeren Zeitraum hinweg, die Lagerstabilität des Torase ids selbst verbessert wird, durch Feuchtigkeits¬ absorption hervorgerufene Veränderungen des Aussehens des Präparates vermieden werden, sowie die Resorption des Torasemids durch den Verdauungskanal noch weiter ver¬ bessert wird und die eine gute Bioverfügbarkeit besitzen. Es wurde gefunden, daß aufgetretene Veränderungen der Formulierungen durch in der Rezeptur vorhandene Mais¬ stärke verursacht werden können. Folglich hat diese Erfindung zum Ziel, derartige Probleme, wie sie bei der herkömmlichen Technik auftreten, zu lösen, und bessere Torasemid-haltige Präparate zur oralen Applikation verfügbar zu machen.
Um die oben beschriebenen Problempunkte zu lösen, wurde mit zahlreichen allgemein verwendeten Komponenten, wie z. B. Bindemitteln und Sprengmitteln, Untersuchungen durch¬ geführt. Es wurde gefunden, daß Torasemid-haltige Zusammensetzungen, die als Bindemittel Hydroxyproypl- cellulose oder Polyvinylpyrrolidon (PVP) , als Sprengmittel Natriu -Croscarmellose oder Crospovidon und übliche formgebende Mittel, wie z. B. Lactose und/oder kristalline Cellulose sowie Schmiermittel, wie z. B. Magnesiumstearat, enthalten, hervorragende Eigenschaften als Präparate zur oralen Applikation besitzen. Darüberhinaus ist der Wirk¬ stoff, das Bindemittel und das Sprengmittel in bestimmten Verhältnissen zueinander einzusetzen.On the other hand, pharmaceutical preparations are desired in which the storage stability of the torase itself is improved, especially when stored over a longer period of time, changes in the appearance of the preparation caused by moisture absorption are avoided, and the absorption of the torasemide is avoided is further improved by the digestive tract and which have good bioavailability. It has been found that changes in the formulations which have occurred can be caused by corn starch present in the formulation. Accordingly, this invention aims to solve such problems as are encountered in the conventional art and to make better torasemide-containing preparations available for oral administration. To solve the problems described above, numerous commonly used components, such as. B. binders and disintegrants, tests carried out. It has been found that torasemide-containing compositions which contain hydroxypropyl cellulose or polyvinylpyrrolidone (PVP) as binders, sodium croscarmellose or crospovidone as disintegrants and customary shaping agents, such as, for. B. lactose and / or crystalline cellulose and lubricants such as. B. magnesium stearate, have excellent properties as preparations for oral administration. In addition, the active ingredient, the binder and the disintegrant are to be used in certain ratios to one another.
Die Erfindung betrifft daher pharmazeutische Zusammen¬ setzungen, die Torasemid oder ein verträgliches Salz davon, ein Bindemittel, ausgewählt aus den Produkten Hydroxypropylcellulose und Polyvinylpyrrolidon (PVP) , ein Sprengmittel, ausgewählt aus den Produkten Natrium- Croscarmellose, Crospovidon, Calcium-Carboxymethyl- cellulose, niedrig substituierte Hydroxypropylcellulose, modifizierte Stärke und Natrium-Carboxymethylstärke und übliche formgebende Mittel sowie Schmiermittel enthalten. Insbesondere betrifft sie pharmazeutische Zusammen¬ setzungen, die 0,1 - 50 Gew. Teile Torasemid, 1 - 10 Gew. Teile Hydroxypropylcellulose und 1 - 20 Gew. Teile Natrium-Croscarmellose und übliche Hilfsstoffe enthält. Eine andere bevorzugte Zusammensetzung enthält 0,1 - 50 Gew. Teile Torasemid, 1 - 10 Gew. Teile Polyvinyl¬ pyrrolidon und 1 - 20 Gew. Teile Crospovidon und übliche Hilfsstoffe.The invention therefore relates to pharmaceutical compositions containing torasemide or a compatible salt thereof, a binder selected from the products hydroxypropyl cellulose and polyvinylpyrrolidone (PVP), a disintegrant selected from the products sodium croscarmellose, crospovidone, calcium carboxymethyl cellulose, contain low-substituted hydroxypropyl cellulose, modified starch and sodium carboxymethyl starch and conventional shaping agents and lubricants. In particular, it relates to pharmaceutical compositions containing 0.1-50 parts by weight of torasemide, 1-10 parts by weight of hydroxypropyl cellulose and 1-20 parts by weight of sodium croscarmellose and customary auxiliaries. Another preferred composition contains 0.1-50 parts by weight of torasemide, 1-10 parts by weight of polyvinyl pyrrolidone and 1-20 parts by weight of crospovidone and conventional auxiliaries.
Bei der vorliegenden Erfindung wird Torasemid, vorzugs¬ weise der Wirkstoff in der Modifikation 1 eingesetzt (siehe EP-A-0,212,537) . Als Verfahren zur Herstellung des Torasemids sei beispielsweise auf das in dem deutschen
Patent DE-B-2,516,025 und in EP-A-0,212,537 beschriebene Verfahren hingewiesen. Die Torasemid-Salze unterliegen keiner besonderen Einschränkung, sofern es sich um phar a- kologisch zulässige Substanzen handelt; als Beispiele lassen sich Salze mit organischen Säuren, wie z. B. Essig¬ säure, Korksäure, Maleinsäure, Fumarsäure, Äpfelsäure, Weinsäure oder Methansulfonsäure, und Salze mit anorganischen Säuren, wie z. B. Salzsäure, Bromwasser¬ stoffsäure, Phosphorsäure oder Schwefelsäure, anführen.In the present invention, torasemide, preferably the active ingredient in modification 1, is used (see EP-A-0,212,537). As a method for producing the Torasemid, for example, that in the German Patent DE-B-2,516,025 and methods described in EP-A-0,212,537. The torasemide salts are not subject to any particular restriction if they are pharmacologically acceptable substances; as examples, salts with organic acids, such as. B. acetic acid, suberic acid, maleic acid, fumaric acid, malic acid, tartaric acid or methanesulfonic acid, and salts with inorganic acids, such as. As hydrochloric acid, hydrobromic acid, phosphoric acid or sulfuric acid.
Bei der vorliegenden Erfindung ist als Bindemittel Hydroxypropylcellulose bevorzugt; insbesondere wurde festgestellt, daß vor allem solche mit hohem Substitu¬ tionsgrad geeignet sind. Der Substitutionsgrad einer solchen hochgradig substituierten Hydroxypropylcellulose beträgt normalerweise 50 - 80 %, vorzugsweise 60 - 70 %, ausgedrückt als Gehalt (%) an Hydroxypropoxyl-Gruppen. Konkret wird handelsübliche Hydroxypropylcellulose verwendet. Weiterhin bevorzugt ist Polyvinylpyrrolidon.In the present invention, hydroxypropyl cellulose is preferred as the binder; in particular, it was found that especially those with a high degree of substitution are suitable. The degree of substitution of such a highly substituted hydroxypropyl cellulose is normally 50-80%, preferably 60-70%, expressed as a content (%) of hydroxypropoxyl groups. Commercial hydroxypropyl cellulose is used specifically. Polyvinylpyrrolidone is also preferred.
Als Sprengmittel werden bevorzugt Natrium-Croscarmellose, Crospovidon, Calcium-Carboxymethylcellulose, niedrig- substituierte Hydroxypropylcellulose, modifizierte Stärke oder Natrium-Carboxymethylstärke eingesetzt. Insbesondere sind Natrium-Croscarmellose oder Crospovidone bevorzugt.Sodium croscarmellose, crospovidone, calcium carboxymethyl cellulose, low-substituted hydroxypropyl cellulose, modified starch or sodium carboxymethyl starch are preferably used as disintegrants. In particular, sodium croscarmellose or crospovidones are preferred.
Unter Natrium-Croscarmellose ist üblicherweise Natrium- Carboxy ethyTcellulose, quervernetzt, zu verstehen, unter Crosprovidon Polyvinylpyrrolidon, quervernetzt. Eine modifizierte Stärke ist vorzugswiese eine in -Stellung konvertierte Stärke.Sodium croscarmellose is usually understood to mean sodium carboxy ethyl cellulose, crosslinked, crosprovidone polyvinylpyrrolidone, crosslinked. A modified starch is preferably a starch converted to the position.
Der pharmazeutischen Zusammensetzung entsprechend dieser Erfindung werden darüber hinaus formgebende Mittel oder Schmiermittel beigemengt werden; als formgebende Mittel werden bevorzugt z. B. Lactose oder kristalline Cellulose
verwendet, und als Schmiermittel wird bevorzugt z. B. Magnesiums earat verwendet.Shaping agents or lubricants will also be added to the pharmaceutical composition according to this invention; as shaping agents are preferred z. B. lactose or crystalline cellulose used, and as a lubricant z. B. magnesium earat used.
Das Mischungsverhältnis der einzelnen Komponenten beträgt bei der pharmazeutischen Zusammensetzung dieser Erfindung normalerweise 0,1 - 50 Teile (hier und im folgenden: Gewichtsteile), bevorzugt 0,5 - 15 Teile, noch bevorzugter 1 - 10 Teile Torasemid; 1 - 10 Teile, bevorzugt 2 - 5 Teile Bindemittel; 1 - 20 Teile, bevorzugt 3 - 15 Teile, noch bevorzugter 5 - 10 Teile Sprengmittel. Die Anteile der anderen Komponenten, die der pharmazeutischen Zusammensetzung dieser Erfindung beigemischt werden, betragen 10 - 150 Teile, bevorzugt 70 - 140 Teile, noch bevorzugter 100 - 130 Teile formgebende Mittel, und 0,1 - 5 Teile, bevorzugt 0,2 - 2 Teile, noch bevorzugter 0,5 - 1,5 Teile Schmiermittel.The mixing ratio of the individual components in the pharmaceutical composition of this invention is normally 0.1-50 parts (here and hereinafter: parts by weight), preferably 0.5-15 parts, more preferably 1-10 parts of torasemide; 1-10 parts, preferably 2-5 parts binder; 1-20 parts, preferably 3-15 parts, more preferably 5-10 parts of disintegrant. The proportions of the other components that are mixed into the pharmaceutical composition of this invention are 10-150 parts, preferably 70-140 parts, more preferably 100-130 parts of shaping agents, and 0.1-5 parts, preferably 0.2-2 Parts, more preferably 0.5-1.5 parts of lubricant.
Die pharmazeutische Zusammensetzung dieser Erfindung wird z. B. als Prophylaktikum bzw. Therapeutiku gegen Hypertonie und Ödeme, oder als Diuretikum idealerweise oral appliziert.The pharmaceutical composition of this invention is e.g. B. as a prophylactic or therapeutic against hypertension and edema, or ideally administered orally as a diuretic.
Die pharmazeutische Zusammensetzung dieser Erfindung wird nach üblichen Verfahren zu einem Pulver, einem Granulat, einer Tablette oder Pellets verarbeitet.The pharmaceutical composition of this invention is processed into a powder, granule, tablet or pellet by conventional methods.
Die Applikation der erfindungsge äßen pharmazeutischen Zusammensetzung schwankt je nach Geschlecht, Körper¬ gewicht, Alter, Krankheitszustand, usw. der Patienten; normalerweise wird bei einem Erwachsenen eine Tagesdosis von ca. 1 - 200 mg Torasemid appliziert, vorzugsweise 1 - 30 mg, aufgeteilt auf ein bis mehrere Male pro Tag.The application of the pharmaceutical composition according to the invention varies depending on the sex, body weight, age, disease state, etc. of the patient; Usually, a daily dose of approximately 1 to 200 mg of torasemide is administered to an adult, preferably 1 to 30 mg, divided into one to several times a day.
Um die vorliegende Erfindung konkret zu erläutern, werden im folgenden Ausführungsbeispiele und Versuchsbeispiele angeführt, wobei diese Erfindung durch diese Beispiele j doch in keiner Weise eingeschränkt wird.
Ausführun sbeispiel 1In order to explain the present invention concretely, exemplary embodiments and test examples are given in the following, but this invention is in no way restricted by these examples. Example 1
40 g Torasemid wurden mit 1.064 g Lactose, 32 g Hydroxy¬ propylcellulose (Gehalt an Hydroxypropoxyl-Gruppen 62 %) und 24 g Natrium-Croscarmellose gemischt, mit Wasser pelletiert, getrocknet und anschließend gesiebt. Zu 466 g dieser Substanz wurden 12,9 g Natrium-Croscarmellose sowie 3,2 g Magnesiumstearat gemischt und das ganze dann durch ein üblicher Verfahren zu Tabletten verarbeitet. Auf diese Weise wurden etwa 2.300 Tabletten mit einem Gehalt von 4 mg Torasemid je Tablette hergestellt.40 g torasemide were mixed with 1,064 g lactose, 32 g hydroxypropyl cellulose (content of hydroxypropoxyl groups 62%) and 24 g sodium croscarmellose, pelleted with water, dried and then sieved. 12.9 g of sodium croscarmellose and 3.2 g of magnesium stearate were mixed with 466 g of this substance and the whole was then processed into tablets by a customary process. About 2,300 tablets containing 4 mg of torasemide per tablet were produced in this way.
Ausfύhrunqsbeispiel 2Exemplary embodiment 2
Unter Verwendung von kristalliner Cellulose anstelle der Lactose wurden entsprechend Ausführungsbeisipiel 1 Tabletten hergestellt.Using crystalline cellulose instead of lactose, 1 tablets were produced in accordance with exemplary embodiment.
Versuchsbeispiel 1Experimental example 1
Entsprechend Ausführungsbeispiel l wurden 7 Präparate mit der in Tabelle 1 angegebenen Zusammensetzung hergestellt. Nr. 1 in der Tabelle ist das Ausführungsbeispiel 1 dieser Erfindung
According to embodiment 1, 7 preparations with the composition given in Table 1 were produced. No. 1 in the table is embodiment 1 of this invention
Tabelle 1Table 1
Die Zahlen geben das Gewicht (mg) der Bestandteile in der Tablette an.The numbers indicate the weight (mg) of the ingredients in the tablet.
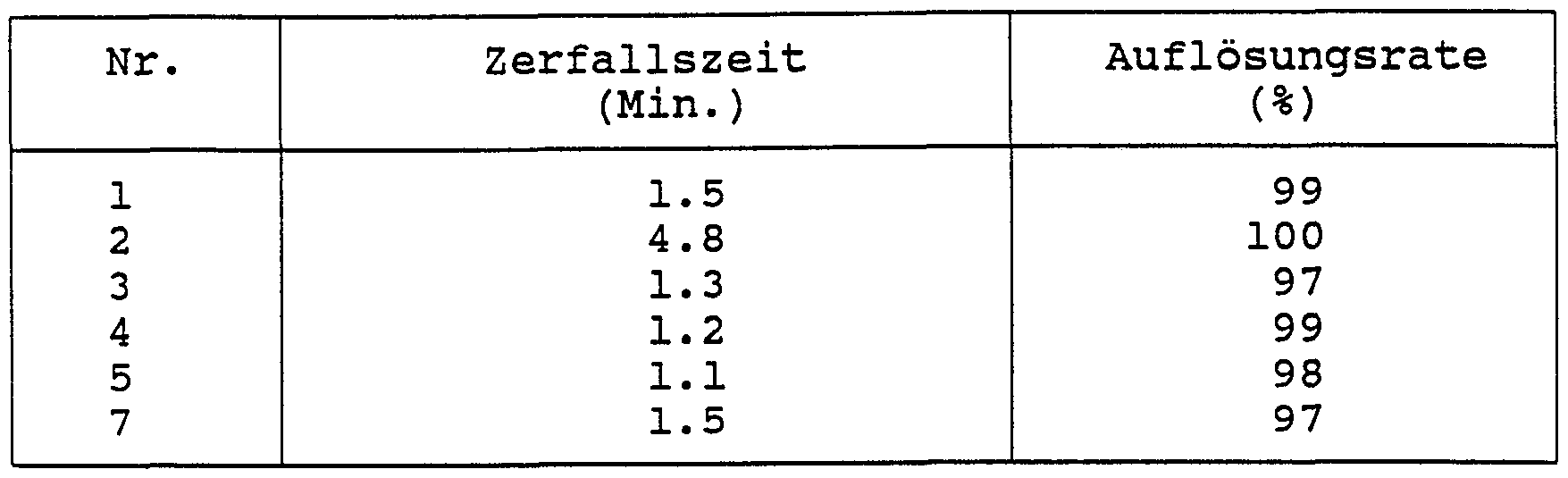
Die Zerfallzeit und die Auflösungsrate (innerhalb von 30 Minuten) der einzelnen Zusammensetzungen wurden ent¬ sprechend des Versuches nach dem Japanischen Arzneibuch, 11. geänderte Ausgabe, bei 37° C in 500 ml durchgeführt; das Ergebnis wird in Tabelle 2 gezeigt.
Tabelle 2The disintegration time and the dissolution rate (within 30 minutes) of the individual compositions were carried out in accordance with the experiment according to the Japanese Pharmacopoeia, 11th revised edition, at 37 ° C. in 500 ml; the result is shown in Table 2. Table 2
Die einzelnen Zusammensetzungen wurden 14 Tage lang bei 40° C und einer relativen Feuchtigkeit von 75 % liegen¬ gelassen und dann die Änderungen des Aussehens, der Härte und des Gewichts untersucht; die Ergebnisse werden in den Tabellen 3 - 5 gezeigt.The individual compositions were left for 14 days at 40 ° C. and a relative humidity of 75% and then the changes in appearance, hardness and weight were examined; the results are shown in Tables 3-5.
Tabelle 3 (Erscheinungsbild)Table 3 (appearance)
-: keine besonderen Veränderungen im Erscheinungsbild +: Glanzverlust an der Tablettenoberfläche-: no special changes in appearance +: loss of gloss on the tablet surface
Tabelle 4 (Härtegrad in N)Table 4 (Degree of hardness in N)
Nachdem die einzelnen Zusammensetzungen einen Monat lang bei 60° C gelagert wurden, wurde der Gehalt an Verun¬ reinigungen untersucht; das Ergebnis wird in Tabelle 6 gezeigt.After the individual compositions had been stored at 60 ° C. for one month, the content of impurities was examined; the result is shown in Table 6.
Tabelle 6Table 6
Versuchsbeispiel 2Experimental example 2
4 mg Torasemid wurden in Form der Zusammensetzung dieser Erfindung (Bsp. 1) Hunden (Körpergewicht 8 - 12 kg, 6 Fälle) oral appliziert; bis zur 24. Stunde betrug die Fläche unter der Kurve der Torasemid-Konzentration im Blut 40,5 +/- 7,8 (/u/ml x hr) .
4 mg of torasemide were administered orally in the form of the composition of this invention (Example 1) to dogs (body weight 8-12 kg, 6 cases); up to the 24th hour the area under the curve of the torasemide concentration in the blood was 40.5 +/- 7.8 (/ u / ml x hr).
Claims
1. Lagerstabile pharmazeutische Zusammensetzung, ent¬ haltend als Wirkstoff Torasemid oder ein verträgliches Salz davon, ein Bindemittel, ausgewählt aus den Produkten Hydroxypropylcellulose und Polyvinyl¬ pyrrolidon (PVP), ein Sprengmittel, ausgewählt aus den Produkten Natrium-Croscarmellose, Crospovidon, Calcium- Carboxymethylcellulose, niedrig substituierte Hydroxy¬ propylcellulose, modifizierte Stärke und Natrium- Carboxymethylstärke und übliche formgebende Mittel sowie Schmiermittel.1. Storage-stable pharmaceutical composition containing as active ingredient torasemide or a compatible salt thereof, a binder selected from the products hydroxypropyl cellulose and polyvinyl pyrrolidone (PVP), a disintegrant selected from the products sodium croscarmellose, crospovidone, calcium carboxymethyl cellulose , low-substituted hydroxypropyl cellulose, modified starch and sodium carboxymethyl starch and conventional shaping agents and lubricants.
2. Pharmazeutische Zusammensetzung gemäß Anspruch 1, dadurch gekennzeichnet, daß das Bindemittel Hydroxy¬ propylcellulose und das Sprengmittel Natrium- Croscarmellose ist.2. Pharmaceutical composition according to claim 1, characterized in that the binder is Hydroxy¬ propyl cellulose and the disintegrant is sodium croscarmellose.
3. Pharmazeutische Zusammensetzung gemäß Anspruch 2, dadurch gekennzeichnet, daß sie 0,1 - 50 Gew. Teile Torasemid, 1 - 10 Gew. Teile Hydroxypropylcellulose und 1 - 20 Gew. Teile Natrium-Croscarmellose enthält.3. Pharmaceutical composition according to claim 2, characterized in that it contains 0.1-50 parts by weight of torasemide, 1-10 parts by weight of hydroxypropyl cellulose and 1-20 parts by weight of sodium croscarmellose.
4. Pharmazeutische Zusammensetzung gemäß Anspruch 1, dadurch gekennzeichnet, daß das Bindemittel Polyvinyl¬ pyrrolidon und das Sprengmittel Crospovidon ist.4. Pharmaceutical composition according to claim 1, characterized in that the binder is polyvinyl pyrrolidone and the disintegrant is crospovidone.
5. Pharmazeutische Zusammensetzung gemäß Anspruch 4, dadurch gekennzeichnet, daß sie 0,1 - 50 Gew. Teile Torasemid, 1 - 10 Gew. Teile Polyvinylpyrrolidon und 1 - 20 Gew. Teile Crospovidon enthält. 5. Pharmaceutical composition according to claim 4, characterized in that it contains 0.1-50 parts by weight of torasemide, 1-10 parts by weight of polyvinylpyrrolidone and 1-20 parts by weight of crospovidone.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP92913781A EP0591363A1 (en) | 1991-06-25 | 1992-06-25 | Pharmaceutical preparations containing torasemid |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP181820/91 | 1991-06-25 | ||
| JP18182091A JP3586471B2 (en) | 1991-06-25 | 1991-06-25 | Torasemide-containing pharmaceutical composition |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO1993000097A1 true WO1993000097A1 (en) | 1993-01-07 |
Family
ID=16107395
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP1992/001437 WO1993000097A1 (en) | 1991-06-25 | 1992-06-25 | Pharmaceutical preparations containing torasemid |
Country Status (3)
| Country | Link |
|---|---|
| EP (1) | EP0591363A1 (en) |
| JP (1) | JP3586471B2 (en) |
| WO (1) | WO1993000097A1 (en) |
Cited By (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP1280534B1 (en) * | 2000-03-17 | 2008-07-16 | Abbott Laboratories | Torasemide-containing storage stable pharmaceutical preparations |
| US8071128B2 (en) | 1996-06-14 | 2011-12-06 | Kyowa Hakko Kirin Co., Ltd. | Intrabuccally rapidly disintegrating tablet and a production method of the tablets |
| US8367111B2 (en) | 2002-12-31 | 2013-02-05 | Aptalis Pharmatech, Inc. | Extended release dosage forms of propranolol hydrochloride |
| US8580313B2 (en) | 2009-12-02 | 2013-11-12 | Aptalis Pharma Limited | Fexofenadine microcapsules and compositions containing them |
| US8747895B2 (en) | 2004-09-13 | 2014-06-10 | Aptalis Pharmatech, Inc. | Orally disintegrating tablets of atomoxetine |
| US9040086B2 (en) | 2001-10-04 | 2015-05-26 | Aptalis Pharmatech, Inc. | Timed, sustained release systems for propranolol |
| US9161918B2 (en) | 2005-05-02 | 2015-10-20 | Adare Pharmaceuticals, Inc. | Timed, pulsatile release systems |
| US9884014B2 (en) | 2004-10-12 | 2018-02-06 | Adare Pharmaceuticals, Inc. | Taste-masked pharmaceutical compositions |
| US10471017B2 (en) | 2004-10-21 | 2019-11-12 | Adare Pharmaceuticals, Inc. | Taste-masked pharmaceutical compositions with gastrosoluble pore-formers |
Families Citing this family (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| ATE322260T1 (en) * | 1996-11-15 | 2006-04-15 | Ajinomoto Kk | NATEGLINIDE PREPARATION IN TABLET FORM |
| WO2003059337A1 (en) * | 2002-01-16 | 2003-07-24 | Kowa Company, Ltd. | Medicinal composition containing 2,2-dichloro-12-(4-chlorophenyl)dodecanoic acid |
| US7109242B2 (en) | 2003-05-23 | 2006-09-19 | Kowa Company, Ltd. | Carboxylic compound and medicine comprising the same |
| ES2244324B1 (en) * | 2004-03-25 | 2006-11-16 | Ferrer Internacional, S.A. | DIURETIC COMPOSITIONS OF PROLONGED RELEASE. |
| JP5617382B2 (en) | 2010-06-28 | 2014-11-05 | トヨタ紡織株式会社 | Intake manifold |
Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0212537A1 (en) * | 1985-08-17 | 1987-03-04 | Roche Diagnostics GmbH | Process for the preparation of a stable modification of torasemide, and medicaments containing torasemide |
-
1991
- 1991-06-25 JP JP18182091A patent/JP3586471B2/en not_active Expired - Lifetime
-
1992
- 1992-06-25 WO PCT/EP1992/001437 patent/WO1993000097A1/en not_active Application Discontinuation
- 1992-06-25 EP EP92913781A patent/EP0591363A1/en not_active Withdrawn
Patent Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0212537A1 (en) * | 1985-08-17 | 1987-03-04 | Roche Diagnostics GmbH | Process for the preparation of a stable modification of torasemide, and medicaments containing torasemide |
Cited By (25)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US8071128B2 (en) | 1996-06-14 | 2011-12-06 | Kyowa Hakko Kirin Co., Ltd. | Intrabuccally rapidly disintegrating tablet and a production method of the tablets |
| US8945618B2 (en) | 1996-06-14 | 2015-02-03 | Kyowa Hakko Kirin Co., Ltd. | Intrabuccally rapidly disintegrating tablet and a production method of the tablets |
| US8956650B2 (en) | 1996-06-14 | 2015-02-17 | Kyowa Hakko Kirin Co., Ltd. | Intrabuccally rapidly disintegrating tablet and a production method of the tablets |
| EP1280534B1 (en) * | 2000-03-17 | 2008-07-16 | Abbott Laboratories | Torasemide-containing storage stable pharmaceutical preparations |
| US9040086B2 (en) | 2001-10-04 | 2015-05-26 | Aptalis Pharmatech, Inc. | Timed, sustained release systems for propranolol |
| US9358214B2 (en) | 2001-10-04 | 2016-06-07 | Adare Pharmaceuticals, Inc. | Timed, sustained release systems for propranolol |
| US8367111B2 (en) | 2002-12-31 | 2013-02-05 | Aptalis Pharmatech, Inc. | Extended release dosage forms of propranolol hydrochloride |
| US8747895B2 (en) | 2004-09-13 | 2014-06-10 | Aptalis Pharmatech, Inc. | Orally disintegrating tablets of atomoxetine |
| US9884014B2 (en) | 2004-10-12 | 2018-02-06 | Adare Pharmaceuticals, Inc. | Taste-masked pharmaceutical compositions |
| US11452689B2 (en) | 2004-10-12 | 2022-09-27 | Adare Pharmaceuticals, Inc. | Taste-masked pharmaceutical compositions |
| US10568832B2 (en) | 2004-10-12 | 2020-02-25 | Adare Pharmaceuticals, Inc. | Taste-masked pharmaceutical compositions |
| US10130580B2 (en) | 2004-10-12 | 2018-11-20 | Adare Pharmaceuticals, Inc. | Taste-masked pharmaceutical compositions |
| US10471017B2 (en) | 2004-10-21 | 2019-11-12 | Adare Pharmaceuticals, Inc. | Taste-masked pharmaceutical compositions with gastrosoluble pore-formers |
| US10952971B2 (en) | 2004-10-21 | 2021-03-23 | Adare Pharmaceuticals, Inc. | Taste-masked pharmaceutical compositions with gastrosoluble pore-formers |
| US9579293B2 (en) | 2005-05-02 | 2017-02-28 | Adare Pharmaceuticals, Inc. | Timed, pulsatile release systems |
| US9566249B2 (en) | 2005-05-02 | 2017-02-14 | Adare Pharmaceuticals, Inc. | Timed, pulsatile release systems |
| US10045946B2 (en) | 2005-05-02 | 2018-08-14 | Adare Pharmaceuticals, Inc. | Timed, pulsatile release systems |
| US9161919B2 (en) | 2005-05-02 | 2015-10-20 | Adare Pharmaceuticals, Inc. | Timed, pulsatile release systems |
| US10500161B2 (en) | 2005-05-02 | 2019-12-10 | Adare Pharmaceuticals, Inc. | Timed, pulsatile release systems |
| US9161918B2 (en) | 2005-05-02 | 2015-10-20 | Adare Pharmaceuticals, Inc. | Timed, pulsatile release systems |
| US11147772B2 (en) | 2005-05-02 | 2021-10-19 | Adare Pharmaceuticals, Inc. | Timed, pulsatile release systems |
| US9233105B2 (en) | 2009-12-02 | 2016-01-12 | Adare Pharmaceuticals S.R.L. | Fexofenadine microcapsules and compositions containing them |
| US10166220B2 (en) | 2009-12-02 | 2019-01-01 | Adare Pharmaceuticals S.R.L. | Fexofenadine microcapsules and compositions containing them |
| US10729682B2 (en) | 2009-12-02 | 2020-08-04 | Adare Pharmaceuticals S.R.L. | Fexofenadine microcapsules and compositions containing them |
| US8580313B2 (en) | 2009-12-02 | 2013-11-12 | Aptalis Pharma Limited | Fexofenadine microcapsules and compositions containing them |
Also Published As
| Publication number | Publication date |
|---|---|
| JPH05952A (en) | 1993-01-08 |
| EP0591363A1 (en) | 1994-04-13 |
| JP3586471B2 (en) | 2004-11-10 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP0128482B1 (en) | Galenic forms of oral antidiabetics and process for their preparation | |
| DE69721749T2 (en) | EPROSARTAND DIHYDRATE AND A METHOD FOR PRODUCING AND FORMULATING IT | |
| EP2477660B1 (en) | Pharmaceutical composition comprising active agents metformin and sitagliptin or vildagliptin | |
| DE3720757A1 (en) | DHP COAT TABLET | |
| EP1128831A1 (en) | Pharmaceutical moxifloxacin preparation | |
| DE69814850T2 (en) | PARACETAMOL CONTAINING SLICKABLE TABLET | |
| WO1993000097A1 (en) | Pharmaceutical preparations containing torasemid | |
| DD224216A5 (en) | PROCESS FOR PREPARING ORAL ANTI-DIABETICALLY ACTUATING PREPARATIONS | |
| DE4001622A1 (en) | ORAL DRUGS OF PIMOBENDAN | |
| EP0722719B1 (en) | Process for preparing flavano-lignan preparations | |
| EP0625355B2 (en) | Tablet with improved bioavailability containing dichloromethylenedephosphonic acid as active ingredient | |
| DE69433012T2 (en) | METHOD FOR PRODUCING ORAL DOSAGE FORMULATIONS CONTAINING DICLOFENAC | |
| DE60030654T2 (en) | Use of lasofoxifen | |
| DE60306747T2 (en) | PERINDOPRIL-CONTAINING PHARMACEUTICAL COMPOSITION WITH ORAL DISPERSION | |
| EP0088098B1 (en) | Composition and method for the preparation of tablets which rapidly disintegrate in an aqueous medium | |
| DE69733752T2 (en) | DRUGS CONTAINING OXAPROCINE SODIUM SALT, CALIUM SALT, OR TRIS (HYDROXYMETHYL) AMINOMETHANE SALT | |
| EP0317855B1 (en) | Pharmaceutical products | |
| DE4423177A1 (en) | Antihyperglycemic drugs | |
| DE2446058A1 (en) | GALENIC PREPARATION FOR ALLOPURINOL CONTINUOUS THERAPY | |
| DE3419128A1 (en) | DIHYDROPYRIDINE PREPARATIONS AND METHOD FOR THE PRODUCTION THEREOF | |
| EP0205865A1 (en) | Pharmaceutical preparations with an antihypertensive and cardioprotective effect | |
| DE69930621T2 (en) | MEDICAL COMPOSITIONS FOR IMMEDIATE RELEASE IN ORAL USE | |
| DE19515972A1 (en) | Controlled release pharmaceutical preparations and process for their preparation | |
| DE3419131A1 (en) | DIHYDROPYRIDINE COMBINATION PREPARATIONS AND METHOD FOR THE PRODUCTION THEREOF | |
| DE19815411A1 (en) | Thermogenesis stimulant comprising moxonidine, useful e.g. for treating hypothermia or promoting weight loss, having no cardiac stimulant side effects |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): JP US |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A1 Designated state(s): AT BE CH DE DK ES FR GB GR IT LU MC NL SE |
|
| DFPE | Request for preliminary examination filed prior to expiration of 19th month from priority date (pct application filed before 20040101) | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 1992913781 Country of ref document: EP |
|
| WWP | Wipo information: published in national office |
Ref document number: 1992913781 Country of ref document: EP |
|
| WWW | Wipo information: withdrawn in national office |
Ref document number: 1992913781 Country of ref document: EP |