Abstract
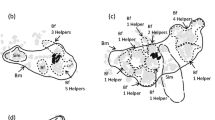
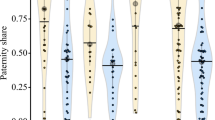
Cooperative breeding systems, where individuals other than parents assist in raising offspring, have been documented in insects, fish, birds, and mammals. Still, the factors driving the evolution of such complex systems are not fully understood. Here, we report a new example of cooperative breeding in the obligate shell-brooding cichlid fish Neolamprologus meeli from Lake Tanganyika. Field observations revealed that dominant males were either monogamous or polygynous, with each mating with one to four dominant females. Dominant females maintain nests containing one to ten empty gastropod shells used as breeding substrates and shelters. Up to four immature subordinates of either sex lived in these nests. They assisted with territory defense and nest maintenance, the frequency of which was not different from that of the dominants. Parentage analyses showed that most subordinates were the offspring of at least one of the breeders, suggesting that juveniles delay dispersal and help in raising their relatives. The relatedness of subordinates to the breeders declined with increasing body size and was significantly higher to female than to male breeders. These patterns could be caused by extra-pair paternity, between-group dispersal of helpers, or sex differences in breeder turnover. Male helpers were larger than female helpers, and six out of eight dispersed individuals were females, suggesting female-biased dispersal. Because N. meeli is phylogenetically distinct from other cooperatively breeding cichlids, these results contribute to a better understanding of cooperative breeding in fishes and to understanding of the evolution of complex social systems in general.
Significance statement
Cooperatively breeding cichlid fishes of Lake Tanganyika are an excellent model for studying the evolution of social complexity because cooperative breeding evolved at least 5–6 times independently in a small phylogenetic group. Here, we provide a new example of cooperative breeding in the cichlid Neolamprologus meeli. Field observations revealed that these fish were either monogamous or polygynous. Subordinates remained in the breeders’ nests and helped with territory defense and nest maintenance. Subordinates of both sexes were unlikely to participate in reproduction because of their immature gonads. Parentage analyses showed that most helpers and juveniles were offspring of the dominant breeders, suggesting that N. meeli has a kin-structured cooperative breeding system. As N. meeli is phylogenetically distinct from all other cooperatively breeding cichlids, these results pose a hitherto undescribed independent evolutionary event, leading to a highly complex social system.
Access this article
We’re sorry, something doesn't seem to be working properly.
Please try refreshing the page. If that doesn't work, please contact support so we can address the problem.





Similar content being viewed by others
Data availability
The dataset generated during the current study is available as supplementary material.
References
Awata S, Heg D, Munehara H, Kohda M (2006a) Testis size depends on social status and the presence of male helpers in the cooperatively breeding cichlid Julidochromis ornatus. Behav Ecol 17:372–379. https://doi.org/10.1093/beheco/arj043
Awata S, Kohda M, Shibata J-Y, Hori M, Heg D (2010) Group structure, nest size and reproductive success in the cooperatively breeding cichlid Julidochromis ornatus: a correlation study. Ethology 116:316–328. https://doi.org/10.1111/j.1439-0310.2009.01735.x
Awata S, Munehara H, Kohda M (2005) Social system and reproduction of helpers in a cooperatively breeding cichlid fish (Julidochromis ornatus) in Lake Tanganyika: field observations and parentage analyses. Behav Ecol Sociobiol 58:506–516. https://doi.org/10.1007/s00265-005-0934-6
Awata S, Takeuchi H, Kohda M (2006b) The effect of body size on mating system and parental roles in a biparental cichlid fish (Julidochromis transcriptus): a preliminary laboratory experiment. J Ethol 24:125–132. https://doi.org/10.1007/s10164-005-0171-5
Balshine S, Leach B, Neat F, Reid H, Taborsky M, Werner N (2001) Correlates of group size in a cooperatively breeding cichlid fish (Neolamprologus pulcher). Behav Ecol Sociobiol 50:134–140. https://doi.org/10.1007/s002650100343
Balshine-Earn S, Neat FC, Reid H, Taborsky M (1998) Paying to stay or paying to breed? Field evidence for direct benefits of helping behavior in a cooperatively breeding fish. Behav Ecol 9:432–438. https://doi.org/10.1093/beheco/9.5.432
Bates D, Mächler M, Bolker BM, Walker SC (2015) Fitting linear mixed-effects models using lme4. J Stat Softw 67:1–48. https://doi.org/10.18637/jss.v067.i01
Bergmüller R, Heg D, Taborsky M (2005) Helpers in a cooperatively breeding cichlid stay and pay or disperse and breed, depending on ecological constraints. Proc R Soc Lond B 272:325–331. https://doi.org/10.1098/rspb.2004.2960
Bergmüller R, Johnstone RA, Russell AF, Bshary R (2007) Integrating cooperative breeding into theoretical concepts of cooperation. Behav Processes 76:61–72. https://doi.org/10.1016/j.beproc.2007.07.001
Bose APH, Windorfer J, Böhm A, Ronco F, Indermaur A, Salzburger W, Jordan A (2020) Structural manipulations of a shelter resource reveal underlying preference functions in a shell-dwelling cichlid fish. Proc R Soc Lond B 287:20200127. https://doi.org/10.1098/rspb.2020.0127
Boulenger GA (1899) Poissons nouveaux du Congo. Cinquième Partie. Cyprins, Silures, Cyprinodontes. Acanthoptérygiens Ann Mus Congo Belge 1:15–127
Brouwer L, Heg D, Taborsky M (2005) Experimental evidence for helper effects in a cooperatively breeding cichlid. Behav Ecol 16:667–673. https://doi.org/10.1093/beheco/ari042
Bruintjes R, Heg-Bachar Z, Heg D (2013) Subordinate removal affects parental investment, but not offspring survival in a cooperative cichlid. Funct Ecol 27:730–738. https://doi.org/10.1111/1365-2435.12088
Carleton KL, Streelman JT, Lee BY, Garnhart N, Kidd M, Kocher TD (2002) Rapid isolation of CA microsatellites from the tilapia genome. Anim Genet 33:140–144. https://doi.org/10.1046/j.1365-2052.2002.00817.x
Clutton-Brock T (2002) Breeding together: kin selection and mutualism in cooperative vertebrates. Science 296:69–72. https://doi.org/10.1126/science.296.5565.69
Clutton-Brock T (2009) Cooperation between non-kin in animal societies. Nature 462:51–57. https://doi.org/10.1038/nature08366
Clutton-Brock TH, O’Riain MJ, Brotherton PNM, Gaynor D, Kansky R, Griffin AS, Manser M (1999) Selfish sentinels in cooperative mammals. Science 284:1640–1644. https://doi.org/10.1126/science.284.5420.1640
Cockburn A (1998) Evolution of helping behavior in cooperatively breeding birds. Annu Rev Ecol Syst 29:141–177. https://doi.org/10.1146/annurev.ecolsys.29.1.141
Cockburn A (2006) Prevalence of different modes of parental care in birds. Proc R Soc Lond B 273:1375–1383. https://doi.org/10.1098/rspb.2005.3458
Cordero PJ, Veiga JP, Moreno J, Parkin DT (2003) Extra-pair paternity in the facultatively polygynous spotless starling, Sturnus unicolor. Behav Ecol Sociobiol 54:1–6. https://doi.org/10.1007/s00265-003-0603-6
Creel SR, Rabenold KN (1994) Inclusive fitness and reproductive strategies in dwarf mongooses. Behav Ecol 5:339–348. https://doi.org/10.1093/beheco/5.3.339
Desjardins JK, Fitzpatrick JL, Stiver KA, Van der Kraak GJ, Balshine S (2008) Costs and benefits of polygyny in the cichlid Neolamprologus pulcher. Anim Behav 75:1771–1779. https://doi.org/10.1016/j.anbehav.2007.09.037
Dey CJ, O’Connor CM, Wilkinson H, Shultz S, Balshine S, Fitzpatrick JL (2017) Direct benefits and evolutionary transitions to complex societies. Nat Ecol Evol 1:0137. https://doi.org/10.1038/s41559-017-0137
Dierkes P, Heg D, Taborsky M, Skubic E, Achmann R (2005) Genetic relatedness in groups is sex-specific and declines with age of helpers in a cooperatively breeding cichlid. Ecol Lett 8:968–975. https://doi.org/10.1111/j.1461-0248.2005.00801.x
Eikenaar C, Richardson DS, Brouwer L, Komdeur J (2007) Parent presence, delayed dispersal, and territory acquisition in the Seychelles warbler. Behav Ecol 18:874–879. https://doi.org/10.1093/beheco/arm047
Eikenaar C, Richardson DS, Brouwer L, Komdeur J (2008) Sex biased natal dispersal in a closed, saturated population of Seychelles warblers Acrocephalus sechellensis. J Avian Biol 39:73–80. https://doi.org/10.1111/j.2008.0908-8857.04124.x
Ekman J, Griesser M (2002) Why offspring delay dispersal: experimental evidence for a role of parental tolerance. Proc R Soc Lond B 269:1709–1713. https://doi.org/10.1098/rspb.2002.2082
Field J, Cronin A, Bridge C (2006) Future fitness and helping in social queues. Nature 441:214–217. https://doi.org/10.1038/nature04560
Frommen JG, Fischer S (2021) Proximate and ultimate mechanisms of cooperation in fishes. In: Kaufman A, Call J, Kaufman J (eds) The Cambridge handbook of animal cognition. Cambridge University Press, pp 272–294
Garvy KA, Hellmann JK, Ligocki IY, Reddon AR, Marsh-Rollo SE, Hamilton IM, Balshine S, O’Connor CM (2015) Sex and social status affect territorial defence in a cooperatively breeding cichlid fish, Neolamprologus savoryi. Hydrobiologia 748:75–85. https://doi.org/10.1007/s10750-014-1899-0
Gaston AJ (1978) The evolution of group territorial behavior and cooperative breeding. Am Nat 112:1091–1100. https://doi.org/10.1086/283348
Goldizen AW, Putland DA, Robertson KA (2002) Dispersal strategies in Tasmanian native hens (Gallinula mortierii). Behav Ecol 13:328–336. https://doi.org/10.1093/beheco/13.3.328
Greenwood PJ (1980) Mating systems, philopatry and dispersal in birds and mammals. Anim Behav 28:1140–1162. https://doi.org/10.1016/S0003-3472(80)80103-5
Griesser M, Drobniak SM, Nakagawa S, Botero CA (2017) Family living sets the stage for cooperative breeding and ecological resilience in birds. PLoS Biol 15:e2000483. https://doi.org/10.1371/journal.pbio.2000483
Griffin AS, Pemberton JM, Brotherton PNM, McIlrath G, Gaynor D, Kansky R, O’Riain J, Clutton-Brock TH (2003) A genetic analysis of breeding success in the cooperative meerkat (Suricata suricatta). Behav Ecol 14:472–480. https://doi.org/10.1093/beheco/arg040
Groenewoud F, Frommen JG, Josi D, Tanaka H, Jungwirth A, Taborsky M (2016) Predation risk drives social complexity in cooperative breeders. P Natl Acad Sci USA 113:4104–4109. https://doi.org/10.1073/pnas.1524178113
Hatchwell BJ, Russell AF, Fowlie MK, Ross DJ (1999) Reproductive success and nest-site selection in a cooperative breeder: effect of experience and a direct benefit of helping. Auk 116:355–363. https://doi.org/10.2307/4089370
Heg D, Bachar Z (2006) Cooperative breeding in the Lake Tanganyika cichlid Julidochromis ornatus. Environ Biol Fishes 76:265–281. https://doi.org/10.1007/s10641-006-9032-5
Heg D, Bachar Z, Brouwer L, Taborsky M (2004) Predation risk is an ecological constraint for helper dispersal in a cooperatively breeding cichlid. Proc R Soc Lond B 271:2367–2374. https://doi.org/10.1098/rspb.2004.2855
Heg D, Bachar Z, Taborsky M (2005) Cooperative breeding and group structure in the Lake Tanganyika cichlid Neolamprologus savoryi. Ethology 111:1017–1043. https://doi.org/10.1111/j.1439-0310.2005.01135.x
Heg D, Rothenberger S, Schürch R (2011) Habitat saturation, benefits of philopatry, relatedness, and the extent of co-operative breeding in a cichlid. Behav Ecol 22:82–92. https://doi.org/10.1093/beheco/arq170
Heg D, Taborsky M (2010) Helper response to experimentally manipulated predation risk in the cooperatively breeding cichlid Neolamprologus pulcher. PLoS One 5:e10784. https://doi.org/10.1371/journal.pone.0010784
Hellmann JK, Sovic MG, Gibbs HL, Reddon AR, O’Connor CM, Ligocki IY, Marsh-Rollo S, Balshine S, Hamilton IM (2016) Within-group relatedness is correlated with colony-level social structure and reproductive sharing in a social fish. Mol Ecol 25:4001–4013. https://doi.org/10.1111/mec.13728
Hogendoorn K, Zammit J (2001) Benefits of cooperative breeding through increased colony survival in an allodapine bee. Insectes Soc 48:392–397. https://doi.org/10.1007/PL00001796
Hori M (1983) Feeding ecology of thirteen species of Lamprologus (Teleostei; Cichlidae) coexisting at a rocky shore of Lake Tanganyika. Physiol Ecol Japan 20:129–149
Hori M, Gashagaza MM, Nshombo M, Kawanabe H (1993) Littoral fish communities in Lake Tanganyika: irreplaceable diversity supported by intricate interactions among species. Conserv Biol 7:657–666. https://doi.org/10.1046/j.1523-1739.1993.07030657.x
Hothorn T, Hornik K, van de Wiel MA, Zeileis A (2008) Implementing a class of permutation tests: the coin package. J Stat Softw 28:1–23. https://doi.org/10.18637/jss.v028.i08
Jordan LA, Maguire SM, Hofmann HA, Kohda M (2016) The social and ecological costs of an ‘over-extended’ phenotype. Proc R Soc Lond B 283:20152359. https://doi.org/10.1098/rspb.2015.2359
Josi D, Flury JM, Reyes-Contreras M, Tanaka H, Taborsky M, Frommen JG (2021a) Sex-specific routes to independent breeding in a polygynous cooperative breeder. Front Ecol Evol 9:750483. https://doi.org/10.3389/fevo.2021.750483
Josi D, Heg D, Takeyama T, Bonfils D, Konovalov DA, Frommen JG, Kohda M, Taborsky M (2021b) Age- and sex-dependent variation in relatedness corresponds to reproductive skew, territory inheritance, and workload in cooperatively breeding cichlids. Evolution 75:2881–2897. https://doi.org/10.1111/evo.14348
Josi D, Taborsky M, Frommen JG (2019) First field evidence for alloparental egg care in cooperatively breeding fish. Ethology 125:164–169. https://doi.org/10.1111/eth.12838
Josi D, Taborsky M, Frommen JG (2020) Investment of group members is contingent on helper number and the presence of young in a cooperative breeder. Anim Behav 160:35–42. https://doi.org/10.1016/j.anbehav.2019.11.013
Jungwirth A, Walker J, Taborsky M (2015) Prospecting precedes dispersal and increases survival chances in cooperatively breeding cichlids. Anim Behav 106:107–114. https://doi.org/10.1016/j.anbehav.2015.05.005
Kalinowski ST, Wagner AP, Taper ML (2006) ML-RELATE: a computer program for maximum likelihood estimation of relatedness and relationship. Mol Ecol Notes 6:576–579. https://doi.org/10.1111/j.1471-8286.2006.01256.x
Kingma SA, Bebbington K, Teunissen N, Peters A, Komdeur J (2021) The evolution of delayed dispersal and different routes to breeding in social birds. Adv Stud Behav 53:163–224
Kingma SA, Santema P, Taborsky M, Komdeur J (2014) Group augmentation and the evolution of cooperation. Trends Ecol Evol 29:476–484. https://doi.org/10.1016/j.tree.2014.05.013
Koenig WD (2017) What drives cooperative breeding? PLoS Biol 15:e2002965. https://doi.org/10.1371/journal.pbio.2002965
Koenig WD, Dickinson JL (2004) Ecology and evolution of cooperative breeding in birds. Cambridge University Press, Cambridge
Koenig WD, Dickinson JL (2016) Cooperative breeding in vertebrates. Cambridge University Press, Cambridge
Koenig WD, Pitelka FA, Carmen WJ, Mumme RL, Stanback MT (1992) The evolution of delayed dispersal in cooperative breeders. Q Rev Biol 67:111–150. https://doi.org/10.1086/417552
Kohda M, Heg D, Makino Y, Takeyama T, Shibata J, Watanabe K, Munehara H, Hori M, Awata S (2009) Living on the wedge: female control of paternity in a cooperatively polyandrous cichlid. Proc R Soc Lond B 276:4207–4214. https://doi.org/10.1098/rspb.2009.1175
Kohler U (1998) Zur Struktur und Evolution des Sozialsystems von Neolamprologus multifasciatus (Cichlidae, Pisces), dem kleinsten Schneckenbuntbarsch des Tanganjikasees. Sharker Verlag, Aachen
Kokko H, Ekman J (2002) Delayed dispersal as a route to breeding: territorial inheritance, safe havens, and ecological constraints. Am Nat 160:468–484. https://doi.org/10.1086/342074
Komdeur J (1996) Influence of helping and breeding experienced on reproductive performance in the Seychelles warbler: a translocation experiment. Behav Ecol 7:326–333. https://doi.org/10.1093/beheco/7.3.326
Komdeur J, Eikenaar C, Brouwer L, Richardson DS (2008) The evolution and ecology of cooperative breeding in vertebrates. Encyclopedia of life sciences. John Wiley & Sons Ltd, Chichester, UK, pp 1–8
Komdeur J, Richardson DS, Hammers M, Eikenaar C, Brouwer L, Kingma SA (2017) The evolution of cooperative breeding in vertebrates. eLS pp 1–11. https://doi.org/10.1002/9780470015902.a0021218.pub2
Konings A (2019) Tanganyika cichlids in their natural habitat, 4th edn. Cichlid Press, El Paso
Kuwamura T (1986) Parental care and mating systems of cichlid fishes in Lake Tanganyika: a preliminary field survey. J Ethol 4:129–146. https://doi.org/10.1007/BF02348115
Kuwamura T (1997) The evolution of parental care and mating systems among Tanganyikan cichlids. In: Kawanabe H, Hori M, Nagoshi M (eds) Fish communities in Lake Tanganyika. Kyoto University Press, Kyoto, pp 57–86
Kuznetsova A, Brockhoff PB, Christensen RHB (2017) lmerTest package: tests in linear mixed effects models. J Stat Softw 82:1–26. https://doi.org/10.18637/jss.v082.i13
Lein E, Jordan A (2021) Studying the evolution of social behaviour in one of Darwin’s Dreamponds: a case for the Lamprologine shell-dwelling cichlids. Hydrobiologia 0123456789https://doi.org/10.1007/s10750-020-04473-x
Lucas JR, Waser PM, Creel SR (1994) Death and disappearance: estimating mortality risks associated with philopatry and dispersal. Behav Ecol 5:135–141. https://doi.org/10.1093/beheco/5.2.135
Lukas D, Clutton-Brock T (2017) Climate and the distribution of cooperative breeding in mammals. R Soc Open Sci 4:160897. https://doi.org/10.1098/rsos.160897
Lukas D, Clutton-Brock T (2012) Cooperative breeding and monogamy in mammalian societies. Proc R Soc Lond B 279:2151–2156. https://doi.org/10.1098/rspb.2011.2468
Mabry KE, Shelley EL, Davis KE, Blumstein DT, van Vuren DH (2013) Social mating system and sex-biased dispersal in mammals and birds: a phylogenetic analysis. PLoS One 8:e57980. https://doi.org/10.1371/journal.pone.0057980
Marshall TC, Slate J, Kruuk LEB, Pemberton JM (1998) Statistical confidence for likelihood-based paternity inference in natural populations. Mol Ecol 7:639–655. https://doi.org/10.1046/j.1365-294x.1998.00374.x
Mulder RA, Dunn PO, Cockburn A, Lazenby-Cohen KA, Howell MJ (1994) Helpers liberate female fairy-wrens from constraints on extra-pair mate choice. Proc R Soc Lond B 255:223–229. https://doi.org/10.1098/rspb.1994.0032
Mulder RA, Langmore NE (1993) Dominant males punish helpers for temporary defection in superb fairy-wrens. Anim Behav 45:830–833. https://doi.org/10.1006/anbe.1993.1100
Nichols HJ, Jordan NR, Jamie GA, Cant MA, Hoffman JI (2012) Fine-scale spatiotemporal patterns of genetic variation reflect budding dispersal coupled with strong natal philopatry in a cooperatively breeding mammal. Mol Ecol 21:5348–5362. https://doi.org/10.1111/mec.12015
Painter JN, Crozier RH, Poiani A, Robertson RJ, Clarke MF (2000) Complex social organization reflects genetic structure and relatedness in the cooperatively breeding bell miner, Manorina melanophrys. Mol Ecol 9:1339–1347. https://doi.org/10.1046/j.1365-294X.2000.01012.x
Peer K, Taborsky M (2007) Delayed dispersal as a potential route to cooperative breeding in ambrosia beetles. Behav Ecol Sociobiol 61:729–739. https://doi.org/10.1007/s00265-006-0303-0
Poll M (1948) Description de Cichlidae nouveaux recueillis par la mission hydrobiologique belge au lac Tanganika (1946–1947). Bull Mus Roy Hist Nat Belgique 24:1–31
R Core Team (2020) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria, http://www.R-project.org. Accessed 22 June 2020
Rasband W (2018) ImageJ. U. S. National Institutes of Health, Bethesda, Maryland, USA. https://imagej.nih.gov/ij/. Accessed 4 May 2020
Reddon AR, O’Connor CM, Marsh-Rollo SE, Balshine S, Gozdowska M, Kulczykowska E (2015) Brain nonapeptide levels are related to social status and affiliative behaviour in a cooperatively breeding cichlid fish. R Soc Open Sci 2:140072. https://doi.org/10.1098/rsos.140072
Rice WR (1989) Analyzing tables of statistical tests. Evolution 43:223–225. https://doi.org/10.2307/2409177
Riehl C (2013) Evolutionary routes to non-kin cooperative breeding in birds. Proc R Soc Lond B 280:20132245. https://doi.org/10.1098/rspb.2013.2245
Ronco F, Büscher HH, Indermaur A, Salzburger W (2020) The taxonomic diversity of the cichlid fish fauna of ancient Lake Tanganyika, East Africa. J Great Lakes Res 46:1067–1078. https://doi.org/10.1016/j.jglr.2019.05.009
Ronco F, Matschiner M, Böhne A et al (2021) Drivers and dynamics of a massive adaptive radiation in cichlid fishes. Nature 589:76–81. https://doi.org/10.1038/s41586-020-2930-4
Rubenstein DR (2016) Superb starlings: cooperation and conflict in an unpredictable environment. In: Koenig WD, Dickinson JL (eds) Cooperative breeding in vertebrates. Cambridge University Press, Cambridge, pp 181–196
Russell AF (2004) Mammals: comparisons and contrasts. In: Koenig WD, Dickinson JL (eds) Ecology and evolution of cooperative breeding in birds. Cambridge University Press, Cambridge, pp 210–227
Russell AF, Langmore NE, Gardner JL, Kilner RM (2008) Maternal investment tactics in superb fairy-wrens. Proc R Soc Lond B 275:29–36. https://doi.org/10.1098/rspb.2007.0821
Saeki T (2022) Studies on cooperative breeding of the cichlid Neolamprologus meeli inhabiting sandy bottom in Lake Tanganyika with a discussion of individual recognition in fish. PhD thesis, Osaka City University, Japan. https://doi.org/10.24544/ocu.20220603-003
Salomon M, Lubin Y (2007) Cooperative breeding increases reproductive success in the social spider Stegodyphus dumicola (Araneae, Eresidae). Behav Ecol Sociobiol 61:1743–1750. https://doi.org/10.1007/s00265-007-0406-2
Sankamethawee W, Hardesty BD, Gale GA (2010) Sex-bias and timing of natal dispersal in cooperatively breeding puff-throated bulbuls Alophoixus pallidus. J Ornithol 151:779–789. https://doi.org/10.1007/s10336-010-0511-2
Sato T, Gashagaza MM (1997) Shell brooding cichlid fishes of Lake Tanganyika. In: Kawanabe H, Hori M, Nagoshi M (eds) Fish communities in Lake Tanganyika. Kyoto University Press, Kyoto, pp 221–240
Satoh S, Nishida Y, Saeki T, Kawasaka K, Kohda M, Awata S (2021) The functional role of sibling aggression and “best of a bad job” strategies in cichlid juveniles. Behav Ecol 32:488–499. https://doi.org/10.1093/beheco/araa150
Satoh S, Saeki T, Kohda M, Awata S (2022) Cooperative breeding in Neolamprologus bifasciatus, a cichlid fish inhabiting the deep reefs of Lake Tanganyika. Ecol Freshw Fish (published online, https://doi.org/10.1111/eff.12658)
Schliewen U, Rassmann K, Markmann M, Markert J, Kocher T, Tautz D (2001) Genetic and ecological divergence of a monophyletic cichlid species pair under fully sympatric conditions in Lake Ejagham, Cameroon. Mol Ecol 10:1471–1488. https://doi.org/10.1046/j.1365-294X.2001.01276.x
Schradin C, Lamprecht J (2000) Female-biased immigration and male peace-keeping in groups of the shell-dwelling cichlid fish Neolamprologus multifasciatus. Behav Ecol Sociobiol 48:236–242. https://doi.org/10.1007/s002650000228
Sopinka NM, Fitzpatrick JL, Desjardins JK, Stiver KA, Marsh-Rollo SE, Balshine S (2009) Liver size reveals social status in the African cichlid Neolamprologus pulcher. J Fish Biol 75:1–16. https://doi.org/10.1111/j.1095-8649.2009.02234.x
Stephens PA, Russell AF, Young AJ, Sutherland WJ, Clutton-Brock TH (2005) Dispersal, eviction, and conflict in meerkats (Suricata suricatta): an evolutionarily stable strategy model. Am Nat 165:120–135. https://doi.org/10.1086/426597
Stiver KA, Fitzpatrick J, Desjardins JK, Balshine S (2006) Sex differences in rates of territory joining and inheritance in a cooperatively breeding cichlid fish. Anim Behav 71:449–456. https://doi.org/10.1016/j.anbehav.2005.06.011
Sunobe T, Munehara H (2003) Mating system and kin relationship between adults and young in the shell-brooding cichlid fish Neolamprologus meeli in Lake Tanganyika. J Ethol 21:87–92. https://doi.org/10.1007/s10164-002-0086-3
Taborsky M (1984) Broodcare helpers in the cichlid fish Lamprologus brichardi: their costs and benefits. Anim Behav 32:1236–1252. https://doi.org/10.1016/S0003-3472(84)80241-9
Taborsky M (1985) Breeder-helper conflict in a cichlid fish with broodcare helpers: an experimental analysis. Behaviour 95:45–75. https://doi.org/10.1163/156853985X00046
Taborsky M (1994) Sneakers, satellites, and helpers: parasitic and cooperative behavior in fish reproduction. Adv Study Behav 23:1–100. https://doi.org/10.1016/S0065-3454(08)60351-4
Taborsky M (2016) Cichlid fishes: A model for the integrative study of social behavior. In: Koenig WD, Dickinson JL (eds) Cooperative breeding in vertebrates: studies of ecology, evolution, and behavior. Cambridge University Press, Cambridge, pp 272–293
Taborsky M, Koblmüller S, Sefc KM, McGee M, Kohda M, Awata S, Hori M, Frommen JG et al (2019) Insufficient data render comparative analyses of the evolution of cooperative breeding mere speculation: a reply to Dey et al. Ethology 125:851–854. https://doi.org/10.1111/eth.12929
Tanaka H, Frommen JG, Engqvist L, Kohda M (2018a) Task-dependent workload adjustment of female breeders in a cooperatively breeding fish. Behav Ecol 29:221–229. https://doi.org/10.1093/beheco/arx149
Tanaka H, Frommen JG, Koblmüller S, Sefc KM, McGee M, Kohda M, Awata S, Hori M, Taborsky M (2018b) Evolutionary transitions to cooperative societies in fishes revisited. Ethology 124:777–789. https://doi.org/10.1111/eth.12813
Tanaka H, Frommen JG, Kohda M (2018c) Helpers increase food abundance in the territory of a cooperatively breeding fish. Behav Ecol Sociobiol 72:51. https://doi.org/10.1007/s00265-018-2450-5
Tanaka H, Frommen JG, Takahashi T, Kohda M (2016) Predation risk promotes delayed dispersal in the cooperatively breeding cichlid Neolamprologus obscurus. Anim Behav 117:51–58. https://doi.org/10.1016/j.anbehav.2016.04.019
Tanaka H, Heg D, Takeshima H, Takeyama T, Awata S, Nishida M, Kohda M (2015) Group composition, relatedness, and dispersal in the cooperatively breeding cichlid Neolamprologus obscurus. Behav Ecol Sociobiol 69:169–181. https://doi.org/10.1007/s00265-014-1830-8
Tanaka H, Kohda M, Frommen JG (2018d) Helpers increase the reproductive success of breeders in the cooperatively breeding cichlid Neolamprologus obscurus. Behav Ecol Sociobiol 72:152. https://doi.org/10.1007/s00265-018-2566-7
Teunissen N, Kingma SA, Fan M, Roast MJ, Peters A (2021) Context-dependent social benefits drive cooperative predator defense in a bird. Curr Biol 31(P4120–4126):E4. https://doi.org/10.1016/j.cub.2021.06.070
Townsend AK, Bowman R, Fitzpatrick JW, Dent M, Lovette IJ (2011) Genetic monogamy across variable demographic landscapes in cooperatively breeding Florida scrub-jays. Behav Ecol 22:464–470. https://doi.org/10.1093/beheco/arq227
van Oppen MJH, Rico C, Deutsch JC, Turner GF, Hewitt GM (1997) Isolation and characterization of microsatellite loci in the cichlid fish Pseudotropheus zebra. Mol Ecol 6:387–388. https://doi.org/10.1111/j.1471-8286.2007.01981.x
Van Wijngaarden C (1995) Distributiepatronen van de Lamprologinen. KU Leuven, Belgium
Venables WN, Ripley BD (2002) Modern applied statistics with S. Springer, New York, New York, NY
von Siemens M (1990) Broodcare or egg cannibalism by parents and helpers in Neolamprologus brichardi (Poll 1986) (Pisces: Cichlidae): a study on behavioural mechanisms. Ethology 84:60–80. https://doi.org/10.1111/j.1439-0310.1990.tb00785.x
Wong M, Balshine S (2011) The evolution of cooperative breeding in the African cichlid fish, Neolamprologus pulcher. Biol Rev 86:511–530. https://doi.org/10.1111/j.1469-185X.2010.00158.x
Yamagishi S, Kohda M (1996) Is the cichlid fish Julidochromis marlieri polyandrous? Ichthyol Res 43:469–471. https://doi.org/10.1007/BF02347645
Zardoya R, Vollmer DM, Craddock C, Streelman JT, Karl S, Meyer A (1996) Evolutionary conservation of microsatellite flanking regions and their use in resolving the phylogeny of cichlid fishes (Pisces: Perciformes). Proc R Soc Lond B 263:1589–1598. https://doi.org/10.1098/rspb.1996.0233
Zeileis A, Hothorn T (2002) Diagnostic checking in regression relationships. R News 2:7–10
Zeileis A, Kleiber C, Jackman S (2008) Regression models for count data in R. J Stat Softw 27:1–25. https://doi.org/10.18637/jss.v027.i08
Zimmermann H, Bose APH, Ziegelbecker A et al (2021) Is biparental defence driven by territory protection, offspring protection or both? Anim Behav 176:43–56. https://doi.org/10.1016/j.anbehav.2021.03.012
Zöttl M, Heg D, Chervet N, Taborsky M (2013) Kinship reduces alloparental care in cooperative cichlids where helpers pay-to-stay. Nat Commun 4:1341. https://doi.org/10.1038/ncomms2344
Acknowledgements
We would like to thank the members of the Maneno Tanganyika Research Team, especially the late Hirokazu Tanaka, who provided constructive feedback during the preliminary field survey of the study, and the staff of the Department of Fisheries, Republic of Zambia, especially Harris Phili, Mbamwai Mbewe, Taylor Banda, and Bumango Musando, for their field support. We thank the anonymous reviewers for their comments on an earlier version of this manuscript and members of the Animal Sociology Laboratory, Osaka City University, for their support throughout the study.
Funding
This study was financially supported by the Japan Society for the Promotion of Science (JSPS) KAKENHI (Grant numbers 20J13379 [to TS], 21K06346 [to SS], 16H05773 and 19H03306 [to MK], and 20KK0168 [to SA]).
Author information
Authors and Affiliations
Contributions
TS, SS, and MK designed the study. TS and SS collected the data. TS, SS, and SA conducted the analyses. TS and SA performed the genetic analyses. TS, SS, and SA wrote the manuscript with significant contributions from JGF. All authors contributed critically to the drafts and approved the manuscript for publication.
Corresponding authors
Ethics declarations
Ethics approval
All experimental protocols were approved by the Animal Care and Use Committee of Osaka City University for Advanced Studies, Japan. Our field observations and sampling in Lake Tanganyika as well as the transportation of specimens for DNA analysis were conducted with permission from the Zambian Ministry of Agriculture, Livestock, and Fisheries, and complied with the current laws in Zambia.
Conflict of interest
The authors declare no competing interests.
Additional information
Communicated by J. Lindström.
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Supplementary file7 (MP4 68660 KB)
Supplementary file8 (MP4 33220 KB)
Rights and permissions
About this article
Cite this article
Saeki, T., Satoh, S., Frommen, J.G. et al. Kin-structured cooperatively breeding groups due to limited dispersal in the obligate shell-brooding cichlid Neolamprologus meeli. Behav Ecol Sociobiol 76, 89 (2022). https://doi.org/10.1007/s00265-022-03201-w
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00265-022-03201-w